Blog
How Molecular Test Orders are Accessioned in Lab with LigoLab's LIS System
March 13, 2026
The Post-Pandemic Pivot in Molecular LIS Diagnostics
Now that healthcare operations have returned to normal following the chaotic COVID-19 pandemic, molecular diagnostics laboratories that invested heavily in PCR testing technology and infrastructure now have the opportunity to pivot and expand their services. With the focus no longer on COVID testing, these laboratories can broaden their test menus and offer a wider range of molecular diagnostic services to their clients.
With modern and flexible molecular lab testing software, particularly advanced medical laboratory information system (LIS) software, organizations can easily configure new analytical workflows and repurpose the capacity that was once dedicated to high-throughput COVID testing. This adaptability enables molecular labs to respond quickly to evolving diagnostic needs and direct their resources to other in-demand molecular LIS testing workflows, including:
- Sexually transmitted infections (STIs)
- Urinary tract infections (UTIs)
- Fungal pathogens
- Respiratory pathogens
- Viral pathogens
What Makes LigoLab's Molecular Lab Software Solution Different?
LigoLab’s all-in-one laboratory information system, which includes integrated laboratory billing and direct-to-consumer lab testing capabilities (via TestDirectly), is an example of a comprehensive and highly configurable diagnostic lab software platform perfectly suited to support today’s modern molecular LIS workflows.
The platform includes powerful modules and automation engines that operate on a shared lab database software and unified infrastructure. This integrated architecture creates a streamlined clinical lab workflow that eliminates the challenges commonly associated with fragmented laboratory software systems, such as duplicate data entry, data inconsistencies, limited automation, and reduced operational visibility.
Several elements of LigoLab’s molecular lab software module distinguish the platform from competing laboratory information systems. One key area is how specimens are accessioned within the molecular LIS workflow. To better understand these advantages, it’s helpful to examine how molecular lab specimens are accessioned in a modern medical LIS environment.
Discover More: The Future of LIS Systems and Personalized Medicine
Step-by-Step: How Test Orders Are Accessioned in LigoLab's Molecular LIS Module
The Molecular LIS module is designed to support every role and department in the lab with flexible, role-based permissions. In the image below, the user is logged in as an Administrator, granting full access to all workflow queues, modules, and departments.
For other users, access can be easily customized, ensuring they only see the specific lab workflow queues relevant to their responsibilities as specimens move from accessioning through processing.
.webp)
Step 1: Logging In and Starting a New Order
You can also see in the same image that the user is in the Ordering Module. To start accessioning a manual order, the user clicks on the New Order workflow queue button.
Also, take note that there is a Remote Orders workflow queue as well. This is where orders from a customer’s electronic health record (EHR) interface or LigoLab Web Connect, LigoLab’s provider portal, first enter the lab with pre-populated fields. The Remote Orders lab workflow queue also supports incoming orders from TestDirectly, LigoLab’s direct-to-consumer lab testing portal.
Step 2: Filling In Order Details
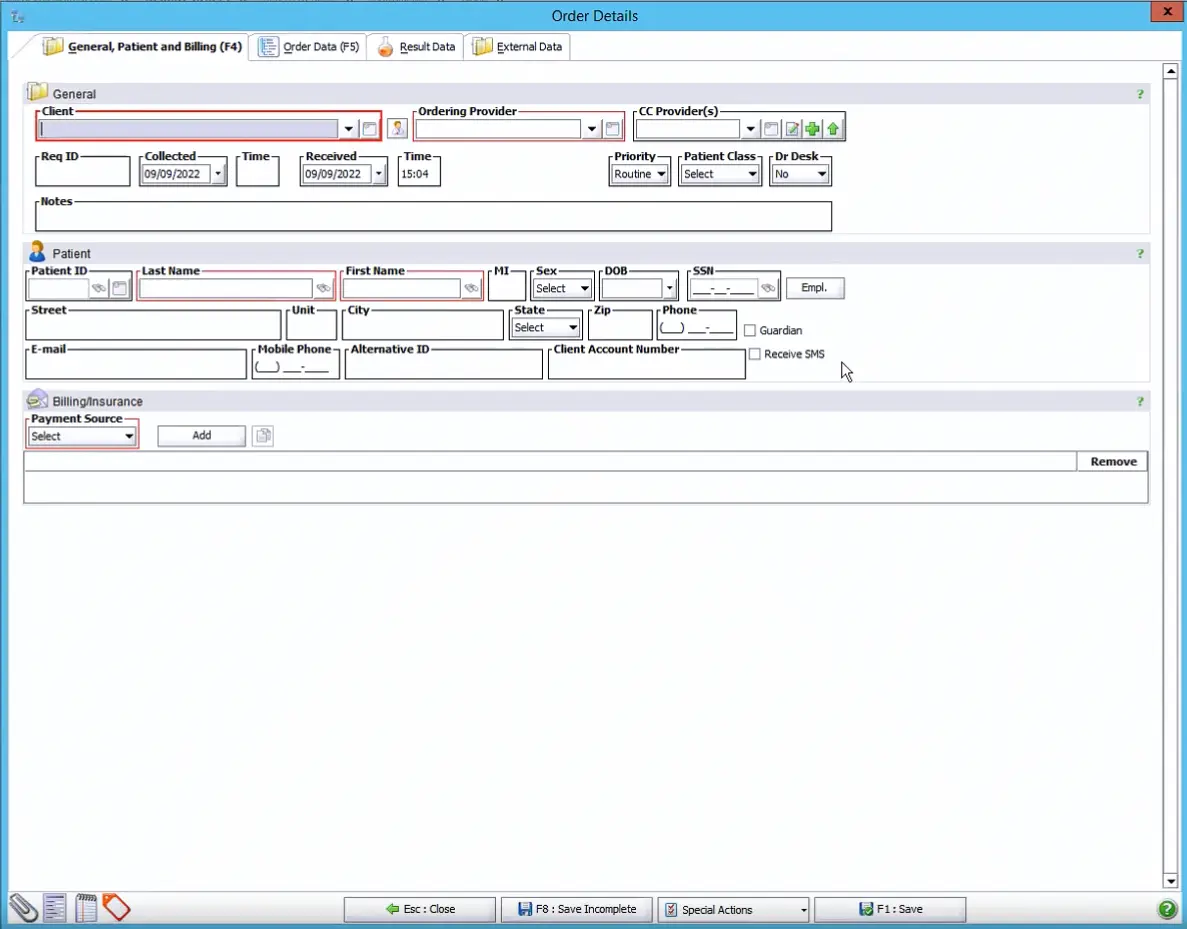
Next, the workflow continues with a manual order entered through the New Order queue. After the order is created, the accessioning user adds the required information in the Order Details window.
As shown in the example, the Order Details window includes three tabs, General, Patient, and Billing, each containing fields that must be completed before the order can be saved. Required fields are clearly highlighted in red and typically include information such as Client, Ordering Provider, Patient Last Name, and Patient First Name. In many laboratory environments, the Payment Source field is also configured as a required entry, ensuring that lab billing information is captured during the accessioning process.
By entering a combination of the patient’s name and date of birth, LigoLab’s molecular lab software module automatically checks for an existing patient profile within the LIS system. If a match is found, a pop-up window will appear, allowing the user to link the current order to the existing profile.
Additionally, the laboratory information system supports real-time address validation through integrations with the United States Postal Service and Whitepages. Users can trigger this feature by clicking the blue icon to the right of the Street field, or the adjacent WP icon, as shown below.
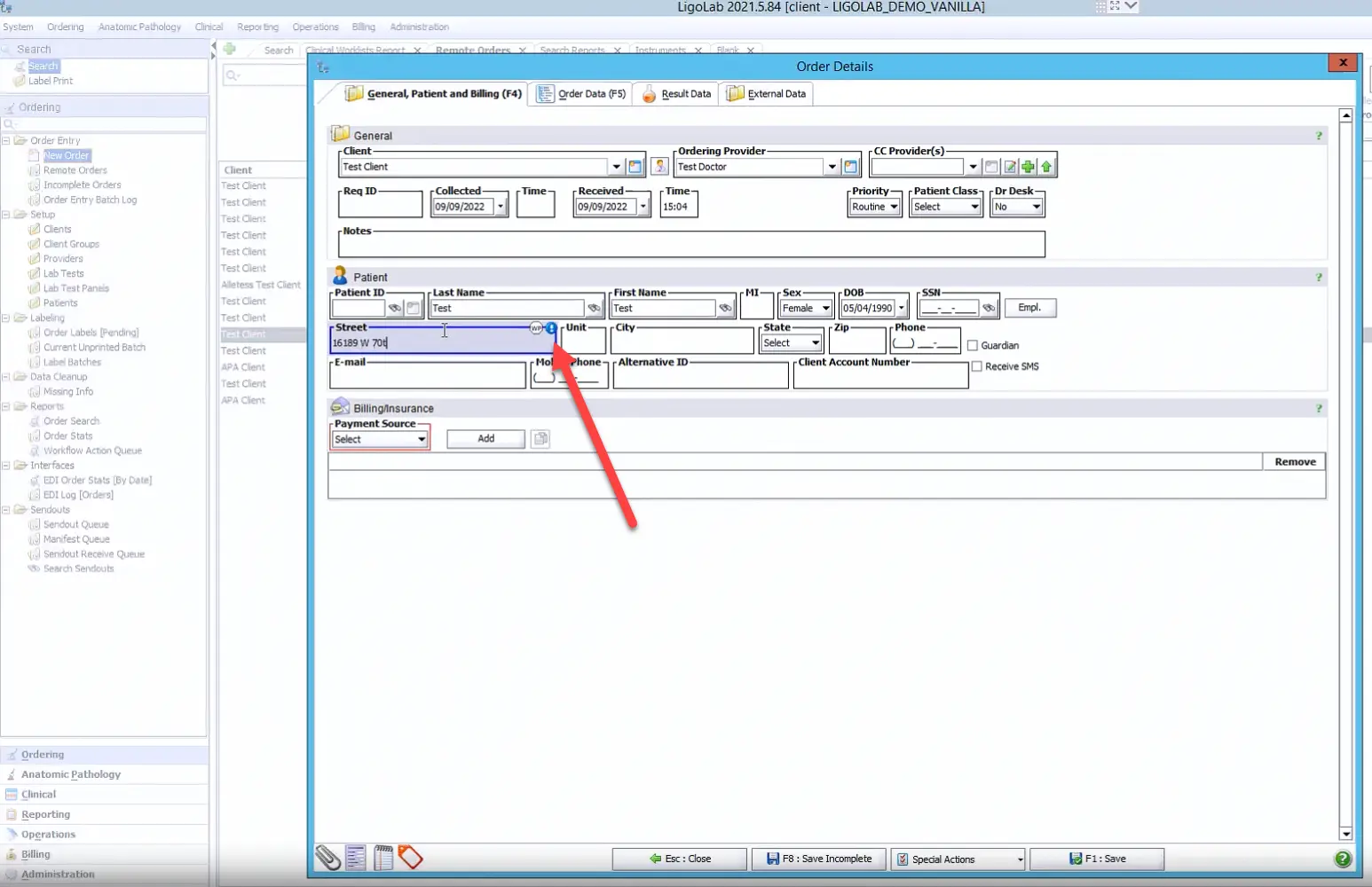
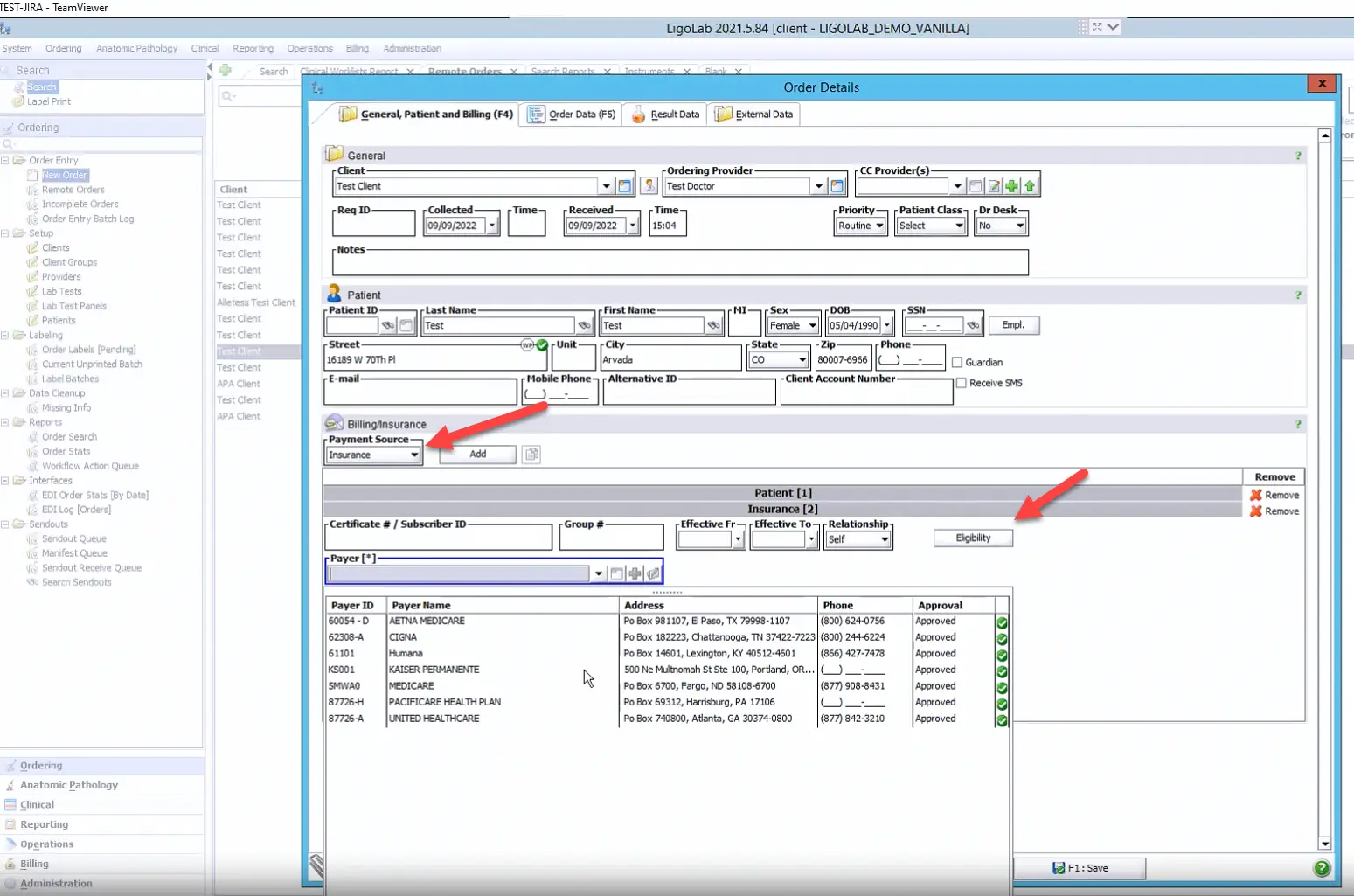
For LigoLab customers who have also licensed the platform’s laboratory billing/lab revenue cycle management module, real-time insurance eligibility checks and insurance discovery validation are also available. Both represent a significant advantage for the lab as they greatly increase the clean claim ratio and dramatically lower the risk of compliance liability.
Next, the user adds the Payment Source. In the example below, insurance has been selected from the Payment Source drop-down menu. Please note that the Insurance Eligibility button has also been highlighted.
Discover More: Comparing LigoLab Informatics Platform with Legacy Laboratory Information System Software
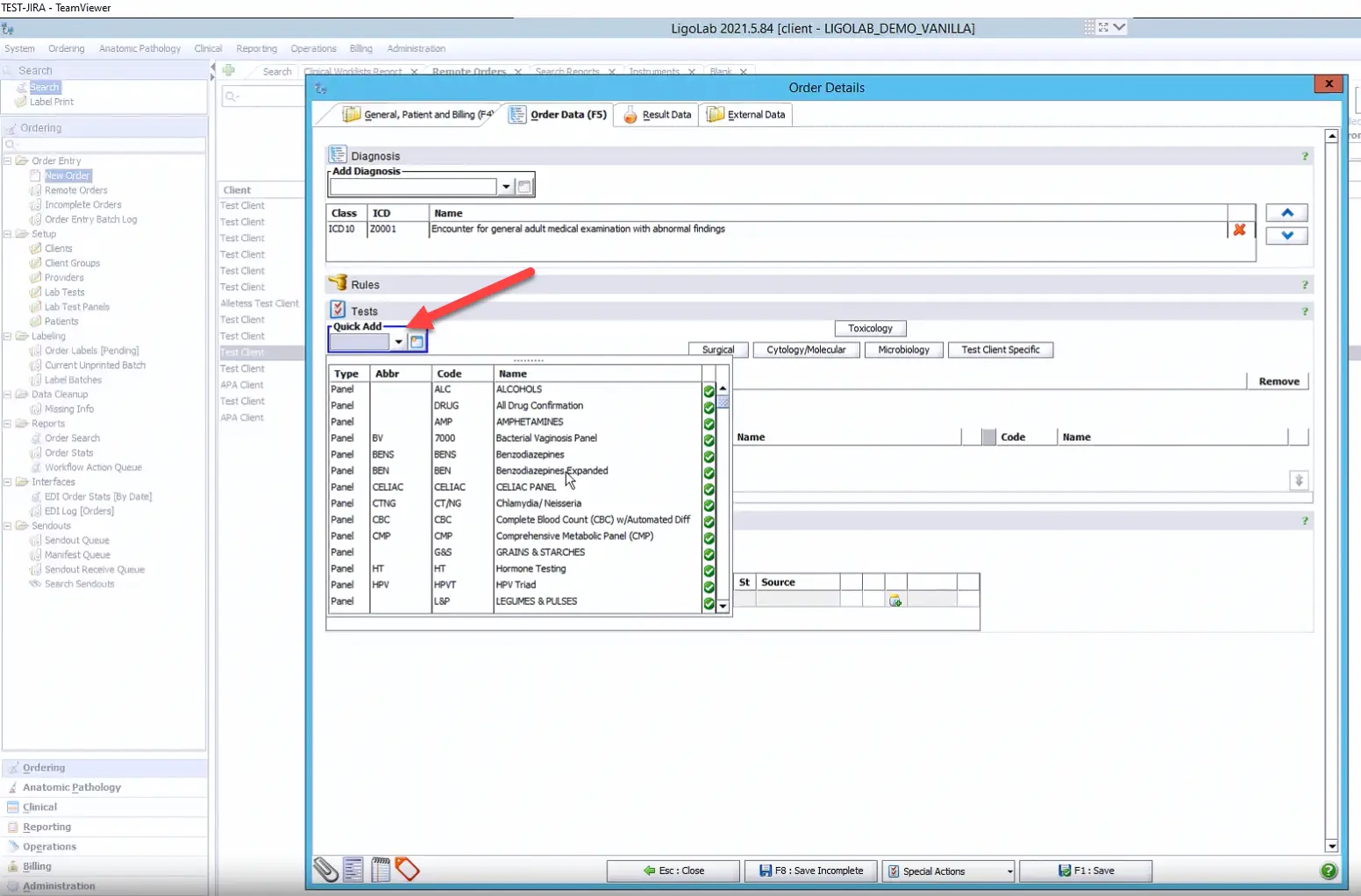
Step 3: Adding ICD-10 Codes, Tests, and Specimens
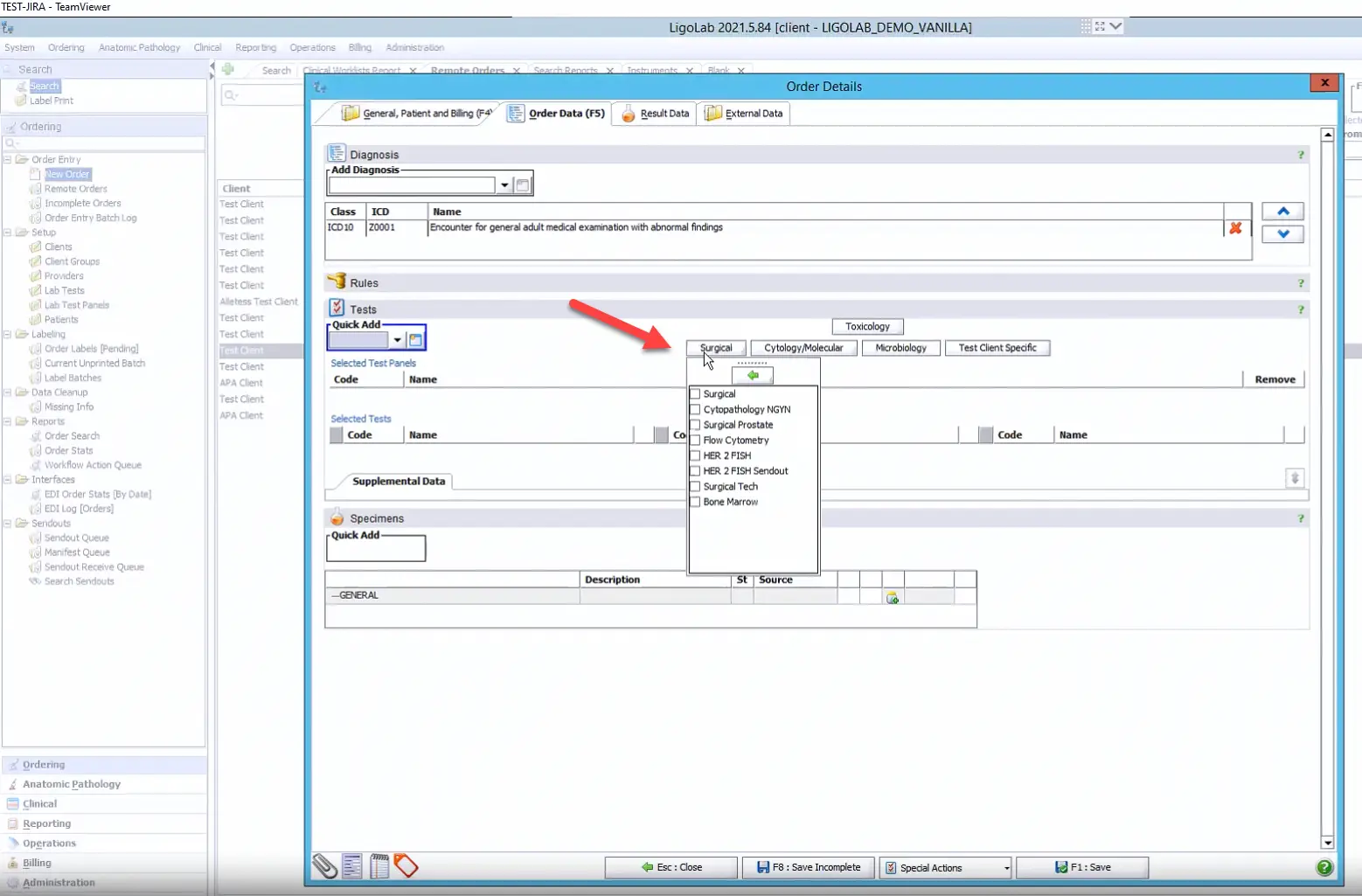
From here, the user goes to the Order Data tab in the Order Details window to add the ICD-10 codes, tests, and specimens. In the example below, the user has clicked on the Quick Add option to see a complete list of the lab’s tests and panels. Please note that this Quick Add functionality can be customized per customer.
There is also a second, shortcut option for frequently ordered tests or panels. Once configured within the laboratory information system, these shortcuts save time for labs with large test menus.
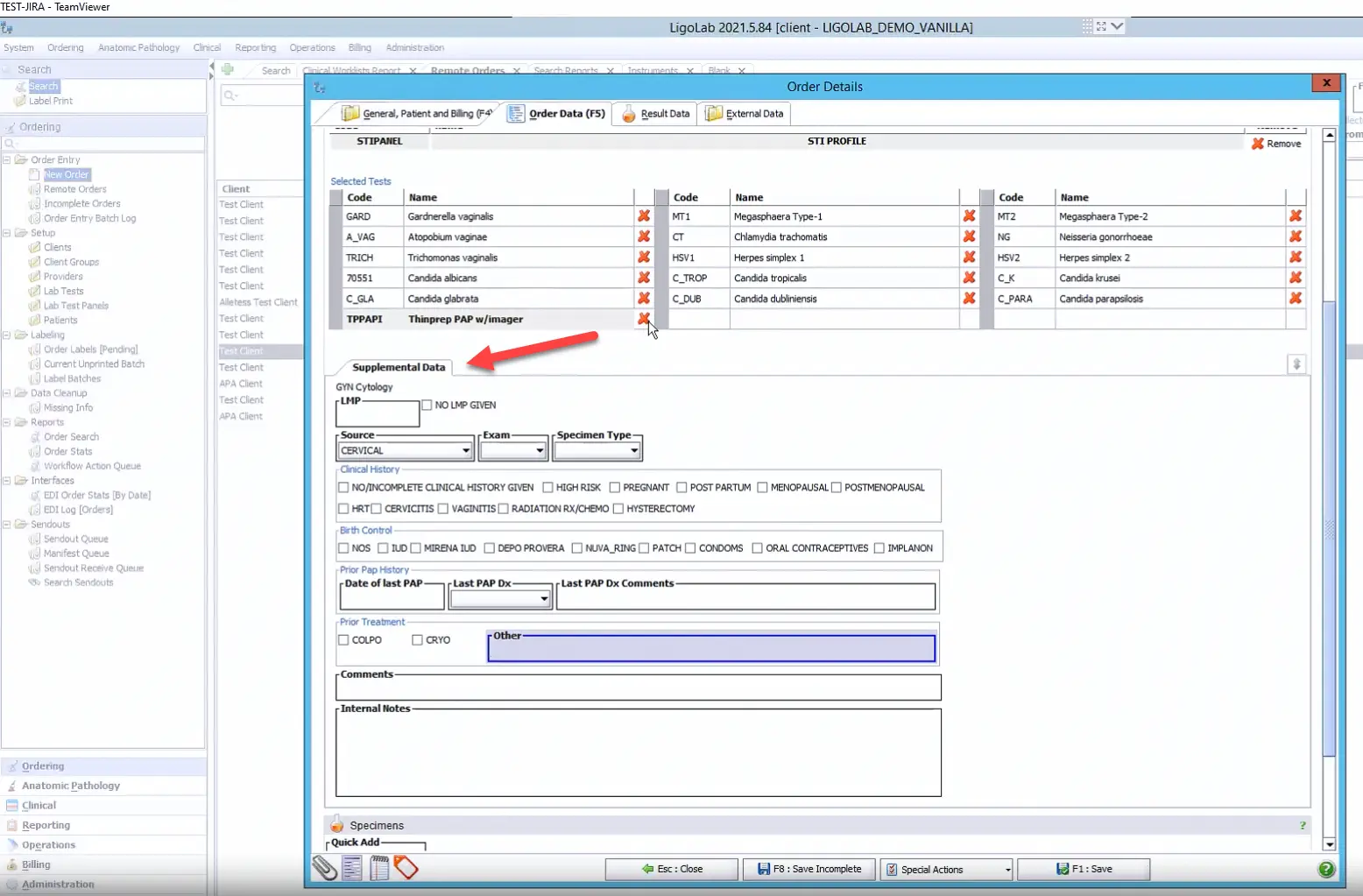
There is also the option to capture more data and add it to the order via the Supplemental Data fields. This area includes checkboxes and options for free text, and all of the info captured here is viewable by users downstream. The data can also be added to the lab report as needed.
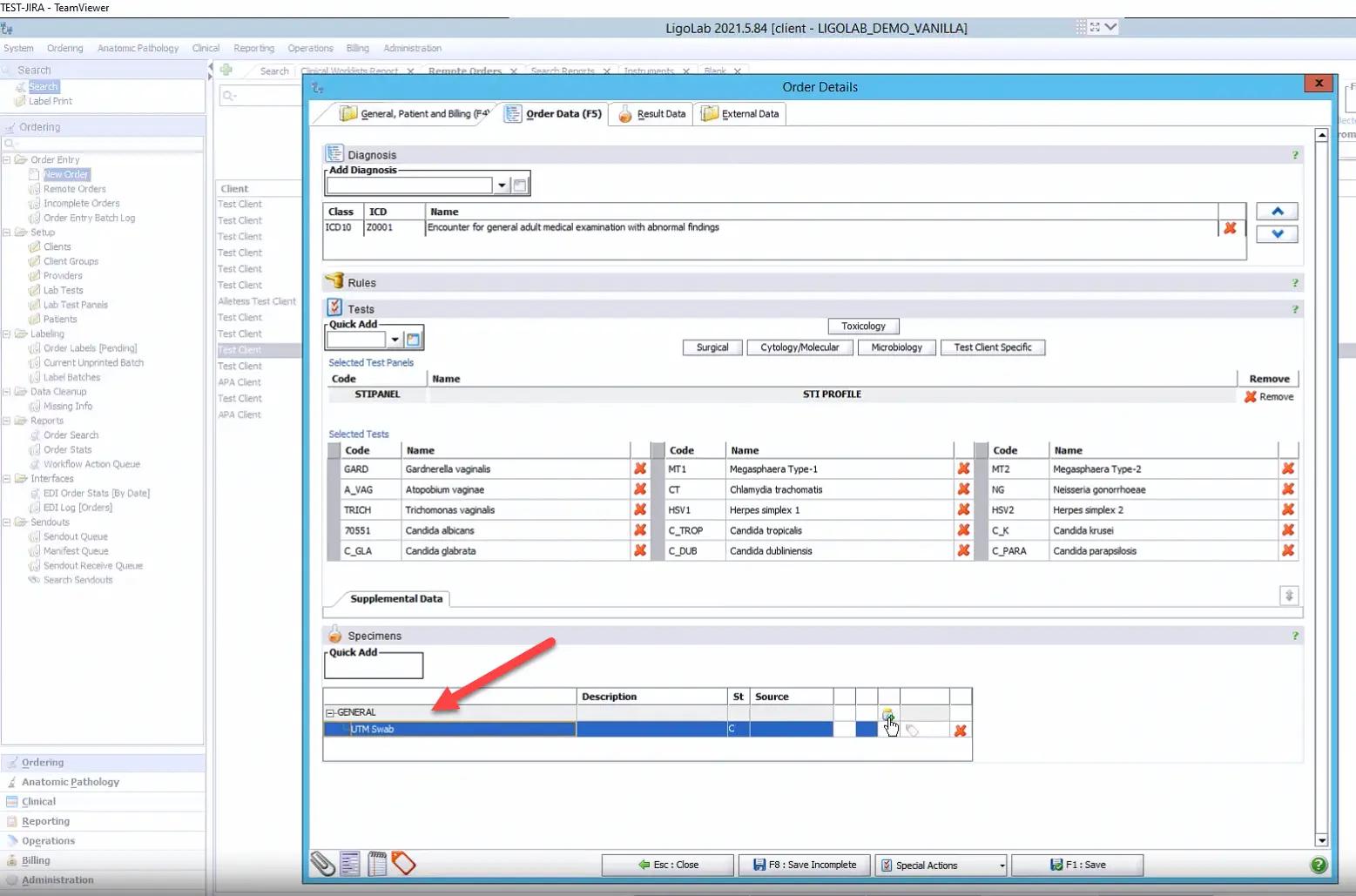
The final section in the Order Data tab is dedicated to specimen entry. It’s a best practice to enter all specimens received with the case under a single order, unless they arrived as separate orders from the customer’s EHR.
The molecular LIS module is built with intelligent logic that can anticipate workflow needs. For instance, it can automatically determine if a specimen should be split into aliquots for downstream processing. Once the order is saved, each specimen is assigned a unique identifier within the LIS software database, ensuring accurate tracking throughout its lifecycle in the lab.
In the case highlighted below, a swab was automatically added to the Specimens section based on the combination of tests selected above it. Users can also add specimens manually, and each specimen has a built-in shortcut that can be typed into the Quick Add field in the Specimens section.
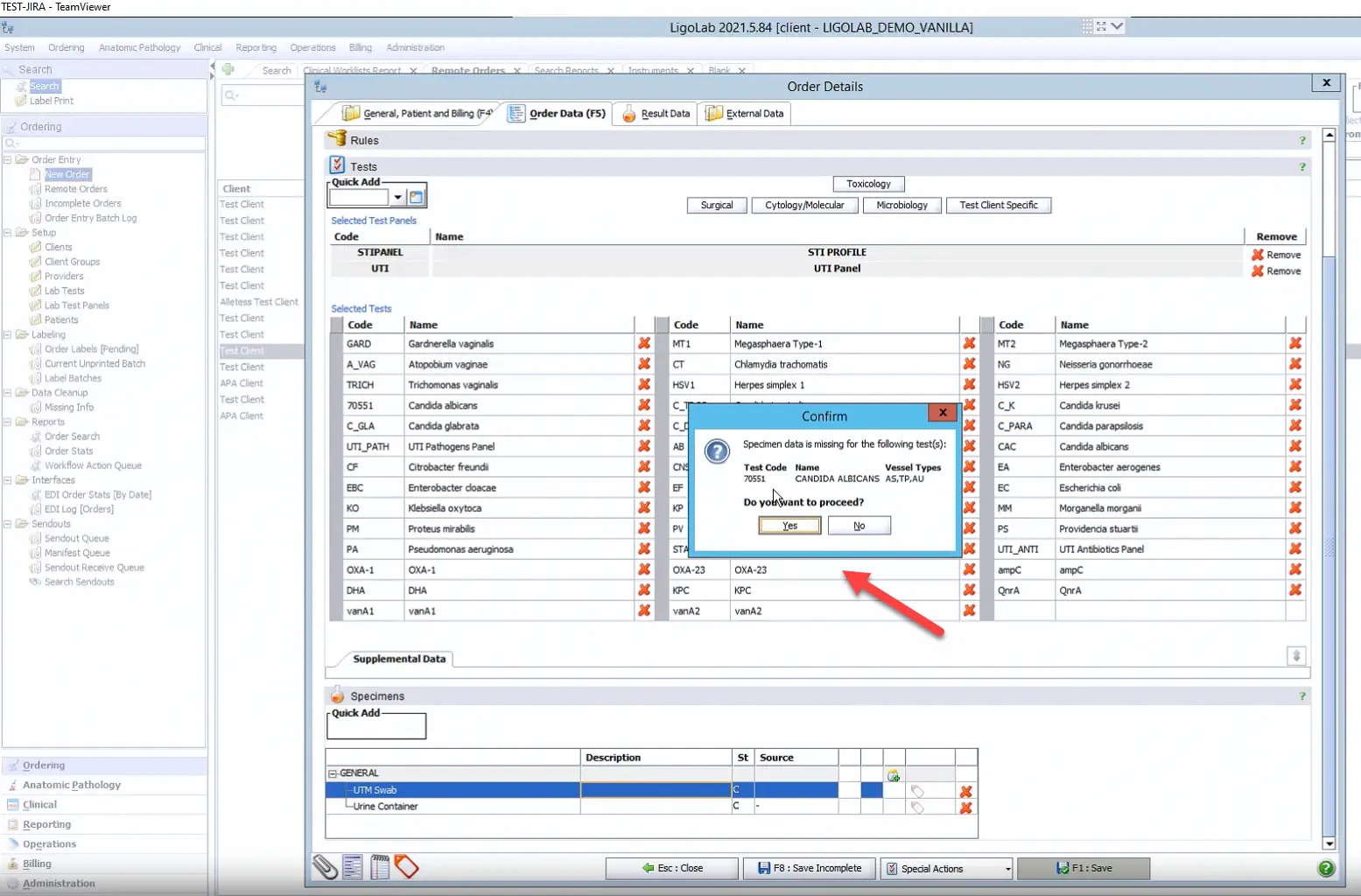
Next, the user clicks the Save button to save the order. If there are any issues with the order, Ligolab’s LIS medical solution will send a notification and ask the user to confirm the order before proceeding.
Step 4: Saving the Order and Triggering Automation
Once the order is saved, an accession number is generated, and each specimen and test is assigned a unique identifier. Tests can then be grouped into reports based on the lab’s preferred workflow. For example, a lab may choose to combine all molecular tests into a single report or generate separate reports for UTI and STI results. These configurations, along with many other variations, are fully supported by the LIS lab platform.
It’s also common for the LIS system’s automation to fire when the order is being saved. At this point, the molecular LIS module checks to make sure that all relevant information has been included within the order (client, patient’s age, what testing was ordered, whether other specimens need to be made, whether subtests need to be added, etc.).
Step 5: Label Printing and Document Attachment
After the order is saved, a Label Print window automatically appears. It's recommended to print one label per specimen, with the specimen identifier embedded in the barcode. This ensures accurate, error-free sample tracking as each specimen is scanned throughout every stage of the clinical lab workflow.
Get Insight: How Specimen Tracking Software Improves Efficiency and Reduces the Chance of Diagnostic Errors
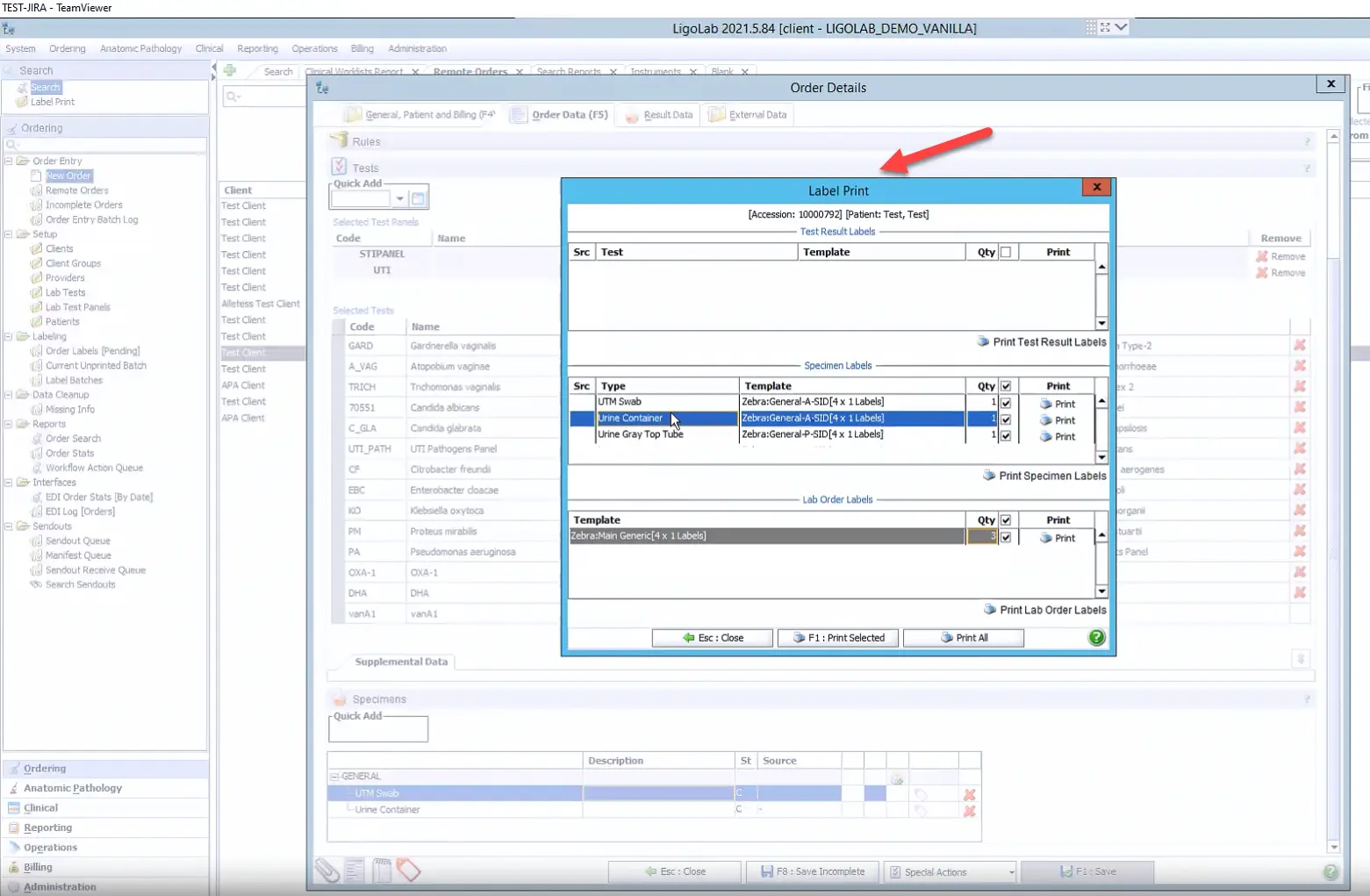
The lower section of the Label Print window (shown above) includes Lab Order Labels. For manually accessioned in-lab orders, it’s recommended to label each page with a barcoded sticker. This enables documents, such as the original requisition, insurance details, and lab billing information, to be scanned in as Lab Order Attachments and easily accessed by any lab technician during specimen processing. Batch scanning these documents later in the workflow is also supported.
Step 6: Ready for the Next Molecular LIS Order
Once the Lab Order Attachments window is closed, the molecular LIS module is ready for the next order. The newly accessioned case and its scheduled tests automatically populate the appropriate workflow queues in the molecular lab software, signaling that processing can begin.
Learn More About LigoLab’s Molecular Lab Testing Software
To discover how LigoLab’s advanced molecular lab testing software can elevate your lab’s performance and why LigoLab Informatics is recognized as one of the best LIS systems available, simply fill out our request-a-demo form to schedule a quick introductory call.
Act Now: Book Your LIS Software Demo Today!
Frequently Asked Questions About Molecular LIS Accessioning
What is accessioning in a molecular diagnostics lab? Accessioning is the process of formally receiving, logging, and preparing a test order for processing. It includes capturing patient demographics, test selections, specimen details, and payment information, and assigning each specimen a unique identifier for tracking throughout the lab workflow.
What is a molecular LIS and why does it matter? A molecular laboratory information system (LIS) is clinical laboratory management software that manages the end-to-end workflow of molecular diagnostic testing, from order entry and accessioning through results reporting and lab billing. A modern molecular LIS like the LigoLab Informatics Platform reduces manual errors, automates key steps, and integrates with EHR systems and lab billing platforms for seamless lab operations.
How does LigoLab's LIS handle accession numbers? When an order is saved in LigoLab's molecular LIS module, an accession number is automatically generated, and each specimen and test is assigned a unique identifier. This enables error-free tracking as specimens move through every stage of the lab workflow.
Can LigoLab's LIS lab solution integrate with EHR systems? Yes. LigoLab's LIS system software supports direct EHR integration, enabling pre-populated orders to automatically flow into the Remote Orders workflow queue, eliminating manual re-entry and accelerating turnaround time from the moment a requisition is submitted.
What makes LigoLab different from other laboratory information systems? Unlike legacy laboratory information systems built from separate modules and lab vendors, LigoLab runs every module, clinical lab, molecular diagnostics, anatomic pathology, and lab billing on a single shared lab database software, and this eliminates typical bottlenecks such as duplicate data entry, data inconsistencies, limited automation, and reduced operational visibility.






